Location: Home >> Detail
TOTAL VIEWS
J Psychiatry Brain Sci. 2026;11(1):e260003. https://doi.org/10.20900/jpbs.20260003
1 Department of Psychiatry, National Clinical Research Center for Mental Disorders, and National Center for Mental Disorders, The Second Xiangya Hospital of Central South University, Changsha 410011, China
2 China National Technology Institute on Mental Disorders & Hunan Key Laboratory of Psychiatry and Mental Health, Mental Health Institute of the Second Xiangya Hospital, Central South University, Changsha 410011, China
* Correspondence: Zongchang Li, Linlin Zhao
Background: Abnormalities in brain structural connectivity have been extensively documented in psychiatric disorders; however, the causal relationships remain largely unclear. Methods: We performed a bidirectional two-sample Mendelian randomization (MR) analysis to investigate the causal relationships between 206 network-level structural connectivity phenotypes (N = 26,333) and 10 psychiatric disorders (N = 14,307 to 1,074,629). Our analysis employed five MR methods, supplemented by a series of sensitivity analyses to validate robustness of the results. Results: The forward MR analysis identified nine network-level structural connectivity phenotypes with causal associations to psychiatric disorders. For instance, decreased white-matter structural connectivity between the left-hemisphere default mode network (DMN) and the right-hemisphere control network (CN), as well as between the left-hemisphere dorsal attention network and the right-hemisphere DMN, was associated with an increased risk of schizophrenia. Furthermore, decreased connectivity between the left and right hemispheres in both the CN and limbic network was correlated with a heightened risk of autism spectrum disorder. The reverse MR analysis revealed a causal relationship where a higher risk of bipolar disorder was associated with decreased white-matter connectivity from the left-hemisphere CN to the putamen. Conclusions: This study represents the first evidence of the causal relationships between network-level structural connectivity and psychiatric disorders, underscoring the critical role of long-range and interhemispheric brain connections. These findings provide new perspectives on the neural mechanisms underlying psychopathology and suggest potential targets for therapeutic interventions and early detection strategies.
GWAS, genome-wide association study; SNP, single-nucleotide polymorphism; IVs, instrumental variables; TS, Tourette’s syndrome; ANX, Anxiety disorders; ASD, Autism spectrum disorder; ANO, Anorexia nervosa; PTSD, Post-traumatic stress disorder; ADHD, Attention deficit hyperactivity disorder; SCZ, Schizophrenia; BD, Bipolar disorder; MDD, Major depressive disorder; IVW, inverse-variance weighted method
Psychiatric disorders are a group of complex conditions characterized by dysfunction in brain activity, affecting nearly one billion people worldwide [1]. These disorders are associated with high incidence, disability, and mortality rates [2], ranking among the top ten global health burdens and posing a significant threat to public health [1]. Despite substantial advancements in the genetics and neurobiology of psychiatric disorders in recent years, their etiology and pathophysiology remain incompletely understood. Structural brain connectivity, which refers to the anatomical connections between different brain regions through neural fiber bundles, is considered vital in the pathogenesis of psychiatric disorders [3,4]. The brain connects different regions through extensive axonal projections and white matter pathways, enabling information exchange between anatomically distinct areas [5]. These large-scale network interactions are critical for cognitive, emotional, and behavioral functions [5,6], making the investigation of structural connectivity abnormalities essential for understanding the underlying pathophysiological mechanisms of psychiatric disorders.
It has been hypothesized that psychiatric disorders can be viewed as dysconnectivity syndromes [7]. Neuroimaging techniques, such as fiber tractography, have enabled researchers to map the connectivity patterns of white matter pathways, measure their integrity, and quantify the connectivity between brain regions [8]. Over the past two decades, neuroimaging studies have provided compelling evidence that psychiatric disorders are closely associated with disruptions in brain network structural connectivity [9–15]. For instance, patients with schizophrenia (SCZ) exhibit impaired integrity of white-matter connections, a significant reduction in the diversity of the topographical distribution of connectivity strength, and considerable heterogeneity in structural connectivity among individuals [11]. In bipolar disorder (BD), cognitive performance involving working memory and problem-solving is associated with changes in the overall structure and connectivity of brain networks [12]. Similarly, in autism spectrum disorder (ASD), widespread reductions in white-matter structural connectivity have been reported [14,16,17], particularly in brain regions associated with restricted behaviors and impaired adaptive functioning, such as the anterior cingulate cortex (ACC) and superior temporal gyrus [16,18].
Traditional research has focused on intra-network structural connectivity, exploring the disruption of connections between regions within the same brain network. However, increasing evidence indicates that the connectivity between different brain networks—i.e., network-level connectivity—may also be important for the development of psychiatric disorders [19–21]. Unlike intra-network connectivity at the brain region level, network-level connectivity provides a more comprehensive perspective, revealing how the brain functions as a dynamic and integrated system [6,22,23]. Network-level connectivity not only reflects the integrity of specific cognitive and emotional processes, but also emphasizes the coordination and communication between these processes [24,25], potentially providing deeper insight into the complex symptoms of psychiatric disorders.
Although a few studies have focused on network-level connectivity abnormalities in psychiatric disorders, most of these have concentrated on changes in functional connectivity [24,26,27], with structural connectivity receiving much less attention. Furthermore, previous observational studies are limited in their ability to establish causal inference due to confounding variables and the possibility of reverse causation. Mendelian randomization (MR) provides a more reliable approach for examining causal relationships by using genetic variants, such as single nucleotide polymorphisms (SNPs), as instrumental variables (IVs) [28]. Since these genetic variants are randomly inherited at conception and unaffected by environmental or behavioral factors, MR provides a natural experiment that can help infer causality [29].
A recent study employing MR has demonstrated causal connections between alterations in resting-state functional connectivity and psychiatric disorders. While structural networks underpin functional connectivity, the causal links between structural brain networks and psychiatric conditions remain poorly understood. In this study, we aim to investigate the causal relationships between network-level structural connectivity phenotypes and ten psychiatric disorders using bidirectional two-sample MR analysis. By utilizing large-scale genome-wide association study (GWAS) data, we examined whether genetic variants associated with network-level structural connectivity influence the risk of psychiatric disorders, and conversely, whether genetic predispositions to psychiatric disorders affect network-level structural brain connectivity. Our study identified ten potential causal relationships, indicating that abnormalities in network-level structural connectivity may have causal effects in disorders such as attention deficit hyperactivity disorder (ADHD), ASD, SCZ, major depressive disorder (MDD), and BD. These findings enhance our understanding of the pivotal role of network-level structural connectivity in the pathogenesis of psychiatric disorders. Furthermore, they offer a more comprehensive perspective on the multidimensional nature of brain dysfunction, proposing new biomarkers and therapeutic strategies for the early detection, prevention, and intervention of these conditions.
Network-level structural connectivity summary statistics were derived from the work of Wainberg et al. [30]. This study performed a GWAS on 206 structural connectivity phenotypes from diffusion magnetic resonance imaging (MRI) tractography data, involving 26,333 participants from the UK Biobank. Each phenotype represents the density of myelinated fibers within or between a pair of large-scale cortical networks, subcortical structures, or hemispheres. Detailed information about these datasets is available in the original publication and Table S1.
Psychiatric DisordersGenetic information for 10 psychiatric disorders was obtained from publicly available GWAS summary data, encompassing Tourette’s syndrome (TS) [31], anxiety disorders (ANX) [32], ASD [33], anorexia nervosa (ANO) [34], post-traumatic stress disorder (PTSD) [35], ADHD [36], SCZ [37], insomnia [38], BD [39] and MDD [40]. The datasets primarily consisted of individuals of European ancestry, with participant counts varying from 14,307 to 1,074,629. Detailed information is provided in Table 1.
Genetic IVs SelectionWe used three strict criteria to select the appropriate genetic instruments for our investigation in order to guarantee reliable causal inference [29]. The instruments must show a strong correlation with the exposure under investigation, not have an independent impact on the study's conclusion, and be impervious to confounding factors. First, we selected SNPs that reached genome-wide significance, adhering to a stringent P value threshold of less than 5 × 10 −8. Second, using the 1000 Genomes Project European dataset as a reference, we used PLINK 1.9 clumping algorithm to find independent SNPs connected to every exposure. With a spatial window of 10,000 kb and a linkage disequilibrium criterion of r2 < 0.001, we carried out clumping under strict guidelines. When SNPs were in linkage disequilibrium, we kept the ones with the lowest P value. Third, we removed SNPs that showed a strong association with the outcome (P < 5 × 10−8). Finally, the SNPs linked to possible confounders were excluded. Four confounders—smoking [41], alcohol consumption [42,43], educational attainment [44,45], and socioeconomic status [46]—were included in the analysis due to previous studies indicating they might have an effect on both structural connectivity and psychiatric disorders. To eliminate SNPs that show a significant connection with these confounders (P < 5 × 10−8) in European participants, we used the NHGRI-EBI GWAS catalog [47] (https://www.ebi.ac.uk/gwas/).
Data HarmonizationTo ensure that the beta values aligned with the same allele, we harmonized the effects of SNPs on both the exposure and the outcome. Additionally, we removed palindromic SNPs with minor allele frequencies close to 0.5, which might cause strand-flipping issues.
Quality Control of Genetic IvsIn order to enhance the dependability of the remaining genetic instruments, we assessed the robustness of selected genetic instruments by calculating the F-statistics using the formula F = R2(N − k − 1)/k (1 − R2), where R2 = β2/(β2 + se2× N) (R 2: the proportion of exposure explained by genetic instruments, N: GWAS sample size for exposure, k: the number of genetic instruments, β: the effect size of the genetic instrument on exposure, se: standard error of the effect size) [48,49]. SNPs with F-statistics less than 10 were eliminated since they were considered weak IVs [50]. The remaining SNPs were subsequently subjected to MR analysis, and their statistical power was determined using Burgess's methodology [51].
Bidirectional Two-Sample MR AnalysesWe used the inverse-variance weighted (IVW) approach with a multiplicative random effects model as the primary method for causal inference in our bidirectional two-sample MR analyses [52]. We also used the weighted median, weighted mode, simple mode, and MR-Egger methods to validate the robustness of our results because of the possible bias in IVW estimations caused by horizontal pleiotropy and invalid genetic instruments. The weighted median method can yield reliable estimates even when as much as 50% of the information is sourced from invalid IVs [53]. The weighted mode method can produce robust estimates even if the most IVs are invalid [54]. The MR-Egger method can offer a more dependable estimation even in cases where all variations are invalid [55]. When there was only one IV available, the Wald ratio was applied. We expanded the aforementioned MR analysis to investigate bidirectional causality between structural connectivity and psychiatric disorders. To account for multiple comparisons, we used the Bonferroni correction in all MR analyses, establishing a statistical significance level of 4.46 × 10−4 (0.05/56/2; 56 denotes the number of structural connectivity measurements, 2 denotes bidirectional MR tests).
Sensitivity AnalysesWe performed several sensitivity analyses to confirm the validity of causal estimates. First, we employed Cochran’s Q test [56] to assess the heterogeneity of individual causal effects within the IVW method. A P-value of less than 0.05 was considered indicative of heterogeneity. Second, we utilized the MR-Pleiotropy Residual Sum and Outlier (MR-PRESSO) [57] to identify pleiotropy (P-value < 0.05) and remove outliers. Third, in order to ascertain whether horizontal pleiotropy exists, we conducted an MR-Egger regression [55]. A directional pleiotropic bias is indicated by an intercept term divergence from zero (P-value < 0.05). Under the assumption of no horizontal pleiotropy, this method evaluates the observed variants by comparing the distances between the variants and the regression line to the expected distances, thereby identifying any anomalies in these variants. Fourth, we performed a leave-one-out analysis to determine if the causal association was influenced by a specific variant [49]. Moreover, we performed the MR Steiger test to assess the validity of the assumption that the exposure causes the outcome.
The study design is summarized in Figure 1. In the initial selection of exposures, we focused on structural connectivity phenotypes for which at least three independent SNPs reached genome-wide significance (P < 5 × 10⁻⁸) in the GWAS. This initial criterion ensured a sufficient pool of genetic instruments for subsequent rigorous filtering. Following this, a total of 56 phenotypes were carried forward for MR analysis. To satisfy the fundamental assumptions of MR, we removed SNPs associated with confounders (Tables S2 and S3). Tables S4 and S5 provide information on the final IVs for significant exposure-outcome pairs in both forward and reverse MR analyses. The F-statistics of all these final IVs exceeded 30, indicating their robustness.
Forward MR of Structural Connectivity on Psychiatric DisordersIn the forward MR analyses, we identified nine statistically significant causal relationships between structural connectivity phenotypes and four psychiatric disorders, including ADHD, ASD, SCZ and MDD (Figure 2 and Table S6). Figure S1 provides scatter plots illustrating genetic associations with psychiatric disorders in relation to genetic associations with structural connectivity.
 Figure 2. Causalities in the forward MR analysis. The forest plot shows the significant causalities estimated using five MR methods. The effect estimates represent the odds ratio (OR) for the risk of psychiatric disorder associated with a 1 standard deviation (s.d.) change in mean structural connectivity phenotype, and the error bars represent 95% confidence intervals (CIs). All statistical tests were two sided. A P value of < 4.46 × 10−4 after Bonferroni correction was considered significant. The pattern diagram on the right shows the brain anatomical regions of the corresponding structural connectivity phenotype.
Figure 2. Causalities in the forward MR analysis. The forest plot shows the significant causalities estimated using five MR methods. The effect estimates represent the odds ratio (OR) for the risk of psychiatric disorder associated with a 1 standard deviation (s.d.) change in mean structural connectivity phenotype, and the error bars represent 95% confidence intervals (CIs). All statistical tests were two sided. A P value of < 4.46 × 10−4 after Bonferroni correction was considered significant. The pattern diagram on the right shows the brain anatomical regions of the corresponding structural connectivity phenotype.
ADHD is a condition related to neurodevelopment that is marked by difficulties with focus, impulsive behavior, and excessive activity levels [58]. Our findings indicated that the white-matter structural connectivity from the left-hemisphere dorsal attention network (DAN) to the right-hemisphere somatomotor network (SMN) was negatively associated with ADHD, with a 1 standard deviation (s.d.) decrease in this connectivity increasing the risk of ADHD by 36% (IVW odds ratio (OR) = 0.64, 95% confidence interval (CI): 0.52 to 0.80, P = 5.09 × 10−5).
Effects of Structural Connectivity on ASDASD is a complex neurodevelopmental disorder defined by fundamental challenges in social interaction and communication, along with repetitive, restrictive, and stereotyped interests, activities, and behaviors [58]. In this study, we identified four structural connectivity phenotypes negatively associated with ASD. Specifically, a 1 s.d. decrease in white-matter structural connectivity between the left-hemisphere visual network (VN) and the caudate nucleus increased ASD risk by 51% (IVW OR = 0.49, 95% CI: 0.35 to 0.68, P = 3.37 × 10−5). Similarly, a 1 s.d. decrease in white-matter structural connectivity between the left and right-hemisphere control networks (CN) increased ASD risk by 42% (IVW OR = 0.58, 95% CI: 0.43 to 0.76, P = 1.40 × 10−4). A 1 s.d. decrease in white-matter structural connectivity between the left and right-hemisphere limbic networks (LN) increased ASD risk by 47% (IVW OR = 0.53, 95% CI: 0.37 to 0.75, P = 3.14 × 10−4). Lastly, a 1 s.d. decrease in white-matter structural connectivity between the left-hemisphere CN and right-hemisphere default mode network (DMN) increased ASD risk by 44% (IVW OR = 0.56, 95% CI: 0.41 to 0.77, P = 3.61 × 10−4).
Effects of Structural Connectivity on SCZSCZ is a severe psychiatric disorder characterized by cognitive disturbances, distorted perception, delusions, difficulties in emotional regulation, altered motor functions, and impaired motivation [59]. Our findings indicated that the white-matter structural connectivity between the left-hemisphere DMN and right-hemisphere CN was negatively associated with SCZ, with a 1 s.d. decrease in this connectivity increasing the risk of SCZ by 33% (IVW OR = 0.67, 95% CI: 0.54 to 0.83, P = 2.41 × 10−4). And the connectivity between the left-hemisphere DAN and right-hemisphere DMN was negatively associated with SCZ, with a 1 s.d. decrease in this connectivity increasing the risk of SCZ by 47% (IVW OR = 0.53, 95% CI: 0.39 to 0.72, P = 3.70 × 10−5).
Effects of Structural Connectivity on MDDMDD is one of the primary causes of disability globally, characterized by physical, cognitive, and emotional symptoms, including fatigue, weight loss, anhedonia, sleep disturbances, and feelings of guilt [60]. Our forward MR analyses indicated that the white-matter structural connectivity from right-hemisphere SMN to amygdala was negatively correlated with the risk of MDD, with a 1 s.d. decrease in this connectivity increasing the risk of MDD by 14% (IVW OR = 0.86, 95% CI: 0.80 to 0.93, P = 2.65 × 10−4). Furthermore, the connectivity from right-hemisphere CN to thalamus was positively related to the risk of MDD, with 1 s.d. increase in this connectivity increasing the risk of MDD by 8% (IVW OR = 1.08, 95% CI: 1.04 to 1.13, P = 1.03 × 10−4).
Reverse MR of Psychiatric Disorders on Structural ConnectivityIn the reverse MR analyses, we found evidence that BD was negatively linked to one structural connectivity phenotype. As shown in Figure 3 and Table S7, a higher risk of BD was associated with decreased white-matter structural connectivity between the left-hemisphere CN and the putamen (IVW β = −0.07, 95% CI: −0.11 to −0.03, P = 3.34 × 10−4).
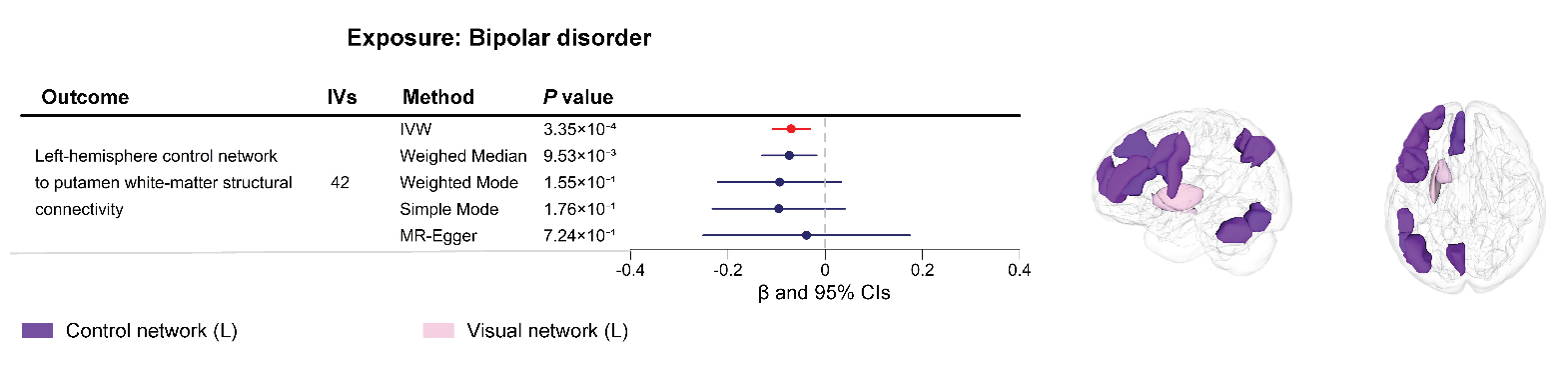 Figure 3. Causalities in the reverse MR analysis. The forest plot shows the significant causalities estimated using five MR methods. The effect estimates represent change in mean structural connectivity phenotype per unit change in disorder, and the error bars represent 95% confidence intervals (CIs). All statistical tests were two sided. A P value of < 4.46 × 10−4 after Bonferroni correction was considered significant. The pattern diagram on the right shows the brain anatomical regions of the corresponding structural connectivity phenotype.
Figure 3. Causalities in the reverse MR analysis. The forest plot shows the significant causalities estimated using five MR methods. The effect estimates represent change in mean structural connectivity phenotype per unit change in disorder, and the error bars represent 95% confidence intervals (CIs). All statistical tests were two sided. A P value of < 4.46 × 10−4 after Bonferroni correction was considered significant. The pattern diagram on the right shows the brain anatomical regions of the corresponding structural connectivity phenotype.
Tables S8 and S9 present the results of sensitivity analyses conducted on the forward and reverse MR analyses. Cochran’s Q test demonstrated no significant heterogeneity among the IVs (P > 0.05). Additionally, the MR-PRESSO test did not reveal any significant horizontal pleiotropy (P > 0.05). Moreover, the MR-Egger intercepts for all associations were close to zero, indicating an absence of significant pleiotropy (P > 0.05). The leave-one-out analyses confirmed that the causal estimates were not dominated by any individual SNP (Figures S1 and S2). Furthermore, the MR Steiger test did not find evidence of reverse causality (Tables S4 and S5).
The directional effects of the estimates from the other four supplementary MR methods were consistent with those obtained using the IVW method. However, applying the same threshold (P < 4.46 × 10−4), the results from the four MR methods did not maintain statistical significance (Figures 2 and 3). This discrepancy could be due to the comparatively lower statistical power of these supplementary MR methods relative to the IVW method [54]. In short, the sensitivity analyses supported the dependability of our proposed causal effects in both the forward and reverse MR results.
This study systematically examined the causal relationship between 206 network-level structural connectivity phenotypes and 10 major psychiatric disorders using the latest GWAS data. By focusing on network-level connectivity, we were able to capture the complex, interconnected nature of brain networks, offering a more comprehensive view than traditional region-specific approaches. In the forward MR analyses, we recognized nine potential causal relationships linking structural connectivity with ADHD, ASD, SCZ, and MDD. The reverse MR analysis identified one putative causal effect of BD on a specific structural connectivity. These results not only highlight the importance of network-level interactions but also provide valuable insights into the neural mechanisms underlying psychopathology.
In our study, forward MR analyses revealed a significant association between decreased white-matter structural connectivity from the left-hemisphere DAN to the right-hemisphere SMN and increased ADHD risk. This supports the hypothesis of impaired functional integration within the attention network and the SMN in ADHD [61,62]. The DAN is consistently engaged in tasks involving verbal and visual working memory and is crucial for top-down, goal-directed attention control [63], whereas the SMN is primarily associated with motor control and sensory processing. Disruption in connectivity between these networks could impair attention regulation and sensorimotor integration, thereby increasing the risk of ADHD. A recent study found that individuals with ADHD have reduced spontaneous neural activity in the DMN, SMN, DAN, and ventral attention networks [64], with decreased DAN connectivity linked to poorer attention performance [64]. Additionally, reduced microstructural integrity within the SMN correlates with increased impulsivity [65]. Impaired inter-network connectivity between the DAN and SMN in children with ADHD has been positively associated with hyperactivity and has a predictive value of 70.4% across cohorts [66]. These findings suggest that abnormal inter-network connectivity between the DAN and SMN may underlie the hyperactivity and impulsivity characteristic of ADHD [66]. Given that the decrease in connectivity in the white matter networks may be the basis for functional deficiencies in gray matter networks [64], decreased white matter integrity could lower the efficiency of information transmission, thereby affecting cognitive and behavioral performance. Our result aligns with previous findings from functional network studies, enhancing the reliability of our conclusions.
This study found a correlation between decreased connectivity in four distinct white matter tracts and heightened susceptibility to ASD, supporting the connectivity deficit theory. This theory posits that ASD is characterized by neurodevelopmental immaturity [67,68] and reduced long-range functional connectivity across large-scale brain networks [68]. Attenuated long-range connectivity between brain regions may act as a key factor in the pathogenesis of ASD. First, we observed that decreased structural connectivity from the left-hemisphere VN to the caudate nucleus is associated with elevated ASD risk. The VN serves as a critical hub for perceptual processing [69,70]. Disrupted connectivity between the VN and the basal ganglia, specifically the caudate nucleus, could impair the integration and reaction to visual cues, especially within social settings. This disruption may influence social cognition and emotional regulation, areas often impaired in those with the disorder [71,72]. Previous research on dynamic brain networks has uncovered altered connectivity in ASD patients involving key regions such as the postcentral gyrus, insula, and caudate nucleus, along with abnormal dynamic interactions between subnetworks like the VN and DMN [73]. We speculate that abnormalities in VN-caudate connectivity may contribute to difficulties in social cognition and emotion recognition. Second, our results show that decreased structural connectivity between the left-hemisphere CN and the right-hemisphere DMN is negatively associated with the risk of ASD. The DMN is essential for self-referential thinking and introspection [74], and deficits in self-awareness and social cognition are particularly pronounced in individuals with ASD [75]. Disruptions in connectivity between the CN and DMN may therefore contribute to these cognitive impairments. Our study also found that decreased interhemispheric connectivity in the CN and LN correlates with increased ASD risk, supporting the notion that individuals with ASD may experience a lack of dynamic cooperation between the cerebral hemispheres [73]. The CN is well-documented for its involvement in executive function, attention control, and cognitive flexibility [76,77], and deficits in these cognitive domains are commonly observed in ASD [78,79]. Our findings highlight the significance of interhemispheric connectivity within the CN for maintaining normal cognitive function, indicating that disruptions in this network may have a potential causal relationship with executive and attentional deficits in ASD. The LN is crucial for emotional regulation, memory, and social behavior [80]. Disrupted development of limbic connectivity is believed to be one neural substrate underlying social impairments in ASD [16]. Research has shown that social functioning deficits in ASD may be linked to abnormal processing of emotional facial expressions. This aberrant processing involves disrupted functional connectivity among brain regions, specifically those linked to the limbic system [81]. Our results provide additional evidence suggesting that weakened limbic connectivity between hemispheres may underlie the social deficits observed in ASD.
In SCZ, we observed that decreased white-matter structural connectivity between the left-hemisphere DMN and the right-hemisphere CN is linked to a higher risk of SCZ. This aligns with prior research indicating that deficits in metacognition in SCZ patients are related to reduced activity in the frontoparietal CN and DMN [82]. Specifically, the anticorrelated balance between these two networks is disrupted during metacognition, with the extent of disruption positively correlating with the severity of negative symptoms [82]. Furthermore, disruptions in DMN-CN functional connectivity have been found to be related to hallucinations in SCZ [83]. Our results further bolster the connection between network imbalance and clinical symptoms in SCZ, suggesting that weakened structural connectivity may be pivotal in this imbalance. We also identified that decreased white-matter structural connectivity from the left-hemisphere DAN to the right-hemisphere DMN is causally related to increased SCZ risk. Previous studies have shown that the DAN and DMN exhibit anticorrelated activity during externally oriented cognition [83,84]. Disruption of this balance through decreased connectivity may lead to cognitive impairments. SCZ patients also exhibit sustained DMN activation during executive function tasks, along with reduced CN activation, suggesting impaired dynamic switching capabilities between networks [83]. Our study further demonstrates that weakened structural connectivity between the DAN and DMN could be a key factor contributing to these cognitive deficits.
This study identified two white-matter structural connections within the right hemisphere associated with MDD risk. We found that decreased connectivity from the right-hemisphere SMN to the amygdala is linked to higher MDD risk. The amygdala, a central region in MDD research [85], plays an important role in emotion processing and response, and its structural and functional alterations are key to understanding the neural basis of MDD [86,87]. Extensive research suggests that amygdala dysfunction is linked to typical MDD symptoms, such as negative self-referential processing, biased negative memory, and attentional bias toward negative stimuli [85,88]. Network analyses combined with machine learning approaches have further revealed disrupted communication between the amygdala and other brain regions in MDD, particularly attenuated connectivity between the SMN and the amygdala [85]. The SMN is involved in motor preparation and control [89], sensory encoding [90], emotion perception [91], and higher cognitive functions, including memory and learning [92]. Studies have consistently shown that diminished functional connectivity between the SMN and amygdala is strongly correlated with motor disturbances in MDD, and our findings further support these studies from a structural connectivity perspective. In addition, we found that increased connectivity between the right-hemisphere CN and the thalamus correlates with higher risk of MDD. The CN is engaged in advanced cognitive processes and regulatory functions [93], while the thalamus acts as a key relay hub for both sensory and affective information, filtering and directing signals to the cerebral cortex [11]. We propose that hyper-connectivity between the CN and thalamus driven by genetic factors may reflect a state of “neural over-control” or inefficient information gating. This excessive top-down control from the CN, rather than supporting adaptive emotional regulation, could over-amplify internally generated thoughts or hinder the thalamus’s ability to habituate to negative stimuli. In turn, this pattern might lead to a cognitive profile marked by excessive rumination and heightened vigilance toward negative cues—two features closely linked to increased susceptibility to MDD. Notably, our MR analysis captures the long-term effects of genetic predisposition over the lifespan. As such, this finding may point to an inherently inefficient compensatory neural wiring pattern: individuals with genetic vulnerability to MDD might have intrinsically stronger CN-thalamus connectivity as an attempt to regulate underlying affective instability. Over time, however, this “over-regulated” baseline state could prove metabolically costly and psychologically draining, ultimately contributing to MDD risk. This perspective moves beyond framing reduced connectivity as purely “protective”, instead highlighting that excessive connectivity in specific regulatory circuits can itself serve as a risk endophenotype for MDD. Future research should explore whether this hyper-connectivity specifically impacts distinct thalamic subnuclei, which are involved in separate cognitive and emotional pathways.
In our reverse MR analyses, we discovered a causal link between increased BD risk and a specific structural connectivity phenotype. Specifically, a higher risk of BD was linked to decreased white-matter structural connectivity from the left-hemisphere CN to the putamen. This finding aligns with current literature, which indicates that, in individuals with BD, the left inferior frontal gyrus shows functional disconnection from a network of areas including the ventrolateral prefrontal gyri, superior temporal gyri, bilateral insulae, and the putamen [94]. In individuals at high risk for BD, low-frequency fluctuation signals in the left putamen were found to be lower compared to healthy controls [95]. Additionally, decreased functional connectivity in regions near the insula and the ACC has also been observed in individuals at high risk for BD [94]. The ACC plays a central role in CN, particularly in monitoring conflict and modifying behavior [96,97]. We speculate that elevated genetic risk for BD may directly contribute to decreased structural connectivity between the CN and the putamen, which in turn leads to impaired functional connectivity between the networks. This may account for the difficulties that BD patients face in monitoring emotional states and cognitive control. However, some findings appear to contradict our results. For instance, impairments in network controllability have been observed in the right inferior frontal and precentral gyri in high-risk BD individuals [98], as well as in a right-lateralized subnetwork that includes the prefrontal cortex, superior temporal pole, putamen, and caudate nucleus [98]. These discrepancies suggest that controllability deficits in BD may also involve right-hemisphere networks, rather than being confined to the left-hemisphere pathways we identified. These differences underscore the intricate nature of the neurobiological mechanisms involved in BD. Further research is necessary to reconcile these findings and thoroughly understand the bilateral characteristics of connectivity deficits associated with the condition.
This study admittedly has some limitations. First, all GWAS involved only European participants to mitigate population stratification bias, potentially limiting the applicability of the results to other ethnic groups. Second, despite rigorous genetic IVs selection procedures, inherent uncertainty in instrumental variable assumptions remains, introducing some ambiguity in causal interpretations. Although attempts were made to exclude SNPs linked to certain confounders during the selection of genetic IVs, it was not possible to account for all potential confounders. Third, some of the identified causal associations were driven by a relatively small number of instrumental variables (as few as two SNPs). Although these IVs exhibited strong F-statistics and the leave-one-out analyses suggested that the estimates were not unduly influenced by any single variant, findings based on few IVs are inherently more susceptible to bias from residual pleiotropy and should be interpreted with caution and require replication in future studies with larger GWAS samples for both brain connectivity and psychiatric disorders. Fourth, MR is a statistical approach that infers potential causality by using genetic associations with both exposure and outcome variables. MR results reflect variations in risk factors over the life course rather than immediate post-intervention effects. Therefore, caution is warranted when translating MR findings to clinical interventions.
This study utilizes the latest GWAS data and MR analyses to uncover causal associations between brain network-level structural connectivity and major psychiatric disorders. The findings reveal significant associations between altered white-matter connectivity and increased risks for ADHD, ASD, SCZ, MDD, and BD, highlighting the pivotal role of network-level structural connectivity impairments in the etiology of these disorders. These results support the connectivity deficit theory in psychiatric disorders and emphasize the importance of long-range and interhemispheric connections in their development. Further studies should explore the mechanisms responsible for the deterioration of these specific network connections and assess their potential as targets for therapeutic intervention.
We utilized genetic datasets derived from previously conducted GWAS studies. The acquisition of institutional ethics committee approvals and study participant informed consent procedures were appropriately addressed in the corresponding original research papers.
Declaration of Helsinki STROBE Reporting GuidelineThis study adhered to the Helsinki Declaration. The Strengthening the Reporting of Observational studies in Epidemiology (STROBE) reporting guideline was followed.
The following supplementary materials are available online, Table S1: GWAS Summary-level data of network-level structural connectivity, Table S2: IVs associated with confounders in forward MR analyses, Table S3: IVs associated with confounders in reverse MR analyses, Table S4: Information of IVs for significant exposure-outcome pairs in forward MR analyses, Table S5: Information of IVs for significant exposure-outcome pairs in reverse MR analyses, Table S6: Significant results of forward MR analyses, Table S7: Significant results of reverse MR analyses, Table S8: Sensitivity analyses for significant exposure-outcome pairs in forward MR analyses, Table S9: Sensitivity analyses for significant exposure-outcome pairs in reverse MR analyses, Figure S1: the scatter plots on the left display the genetic associations between exposure (x-axis) and outcome (y-axis), and the leave-one-out analysis plots of inverse-variance weighted (IVW) results between exposure and outcome are shown on the right, Figure S2: (a) the scatter plots on the left display the genetic associations between bipolar disorder (BD) (x-axis) and left-hemisphere control netwrok to putamen white-matter structural connectivity (GCST90302754) (y-axis). (b) the leave-one-out analysis plots of inverse-variance weighted (IVW) results between BD and GCST90302754 are shown on the right.
The GWAS for network-level structural connectivity can be obtained through the European Bioinformatics Institute GWAS Catalog (https://www.ebi.ac.uk/gwas) under accession numbers GCST90302648 through GCST90302853. The GWAS summary statistics for ADHD, ANO, ANX, ASD, BD, PTSD, SCZ, and TS are available for download at https://pgc.unc.edu/for-researchers/download-results/. The GWAS summary statistics for MDD can be available for download at https://ipsych.dk/en/research/downloads/. The GWAS summary statistics for insomnia can be available for download at https://ctg.cncr.nl/software/summary_statistics/.
LLZ was primarily responsible for the statistical analysis and interpretation of the data, and drafted the initial version of the manuscript. QL performed data collection. AJL prepared the figures. XGC revised the manuscript. ZCL conceptualized and designed the study, provided funding, and participated in manuscript revision. All authors have approved the final manuscript.
The authors declare that there is no conflict of interest.
This study is supported by the National Natural Science Foundation of China (Grant No. 82101576 (ZCL)), Science and Technology Innovation Program of Hunan Province (Grant No. 2022RC1040 (ZCL)), and the Natural Science Foundation of Hunan Province of China (Grant No.2022JJ40695(ZCL)).
We would like to express our gratitude to the research teams from the Psychiatric Genomics Consortium, the UK Biobank, iPSYCH, and Complex Trait Genetics Laboratory for openly sharing their results. We also appreciate the support and collaboration from our colleagues and institutions.
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
40.
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
51.
52.
53.
54.
55.
56.
57.
58.
59.
60.
61.
62.
63.
64.
65.
66.
67.
68.
69.
70.
71.
72.
73.
74.
75.
76.
77.
78.
79.
80.
81.
82.
83.
84.
85.
86.
87.
88.
89.
90.
91.
92.
93.
94.
95.
96.
97.
98.
Liao A, Liu Q, Li Z, Zhao L. Mendelian Randomization Analyses Support Causal Relationships Between Brain Network-Level Structural Connectivity and Risk of Psychiatric Disorders. J Psychiatry Brain Sci. 2026;11(1):e260003. https://doi.org/10.20900/jpbs.20260003.

Copyright © Hapres Co., Ltd. Privacy Policy | Terms and Conditions